
Silver Sulfate (Ag₂SO₄) is an inorganic silver salt composed of silver ions (Ag⁺) and sulfate ions (SO₄²⁻). It appears as a white crystalline solid and is moderately soluble in water compared to other silver salts such as silver chloride.
Silver Sulfate is primarily used in analytical chemistry, electrochemical applications, and as a precursor in the synthesis of other silver-based compounds. Due to its ability to release silver ions, it also exhibits antimicrobial properties, making it relevant in specialized pharmaceutical and laboratory applications.
CAS No.: 10294-26-5
Synonyms: Silver(I) sulfate; Disilver sulfate; Ag₂SO; Argentous sulfate; sulfato de prata; sulfato de plata; Argentum sulfate; Sulfate d’argent
| Physical Properties | |
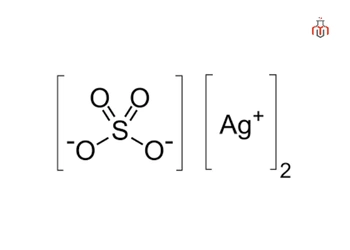
| Chemical Formula | Ag₂SO₄ |
| IUPAC Name | Silver(I) sulfate |
| Molecular Weight | 311.80 g/mol |
| Solubility | Moderately soluble in water (~8 g/L at 25°C) |
| Boiling Point | 1,085 °C |
| Melting Point | 652.2–660 °C |
| Chemical Properties | |
| Appearance | White to gray crystalline powder |
| Odor | Odorless |
| Density | 5.45 g/cm3 (25 °C) |
Silver Sulfate is primarily utilized as a controlled source of silver ions in laboratory, analytical, and specialized industrial applications. Its moderate solubility and stability make it suitable for systems where a regulated release of Ag⁺ ions is required.
| Pictograms : |   |
| Hazard Statements : | H318 Causes serious eye damage |
| Precautionary statements : | Precautionary statements – prevention P273 Avoid release to the environment P280 Wear protective gloves/protective clothing/eye protection/face protection/hearing protection/… Precautionary statements – response P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing P310 Immediately call a POISON CENTER/doctor P391 Collect spillage |
Silver Sulfate (Ag₂SO₄) is an inorganic silver salt composed of silver ions (Ag⁺) and sulfate ions (SO₄²⁻). It appears as a white crystalline solid and is moderately soluble in water compared to other silver salts such as silver chloride.
Silver Sulfate is primarily used in analytical chemistry, electrochemical applications, and as a precursor in the synthesis of other silver-based compounds. Due to its ability to release silver ions, it also exhibits antimicrobial properties, making it relevant in specialized pharmaceutical and laboratory applications.

CAS No.: 10294-26-5
Synonyms: Silver(I) sulfate; Disilver sulfate; Ag₂SO; Argentous sulfate; sulfato de prata; sulfato de plata; Argentum sulfate; Sulfate d’argent
| Physical Properties | |
| Chemical Formula | Ag₂SO₄ |
| IUPAC Name | Silver(I) sulfate |
| Molecular Weight | 311.80 g/mol |
| Solubility | Moderately soluble in water (~8 g/L at 25°C) |
| Boiling Point | 1,085 °C |
| Melting Point | 652.2–660 °C |
| Chemical Properties | |
| Appearance | White to gray crystalline powder |
| Odor | Odorless |
| Density | 5.45 g/cm3 (25 °C) |
Silver Sulfate is primarily utilized as a controlled source of silver ions in laboratory, analytical, and specialized industrial applications. Its moderate solubility and stability make it suitable for systems where a regulated release of Ag⁺ ions is required.
| Pictograms : |   |
| Hazard Statements : | H318 Causes serious eye damage |
| Precautionary statements : | Precautionary statements – prevention P273 Avoid release to the environment P280 Wear protective gloves/protective clothing/eye protection/face protection/hearing protection/… Precautionary statements – response P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing P310 Immediately call a POISON CENTER/doctor P391 Collect spillage |
Silver sulfate is mainly used in analytical chemistry, electrochemical processes, and as a precursor for synthesizing other silver compounds. It is also valued for its antimicrobial properties in controlled applications.
Silver sulfate can be irritating to the eyes, and direct exposure should be avoided. It is advisable to use appropriate safety measures, such as protective eyewear, when handling it.
Silver sulfate is mostly used as a laboratory reagent, an electrochemical material, and a source of silver ions in chemical reactions. It also has limited antimicrobial applications.
Silver sulfate is commonly known as argentous sulfate or disilver sulfate. It is also referred to as silver(I) sulfate or sulfuric acid, disilver(I) salt, used as a reagent in chemical analysis, particularly in laboratory testing and organic synthesis, and also finds application in electrochemistry and water quality analysis.
Silver sulfate solution is formed by dissolving a small amount of silver sulfate in water, resulting in a saturated or near-saturated solution due to its low solubility.
Silver sulfate has a molar solubility in water at 25 °C of approximately 1.4 × 10⁻² mol/L, based on its solubility product (Ksp ≈ 1.2 × 10⁻⁵), indicating relatively low solubility in water.