
Silver citrate is a silver salt of citric acid, stabilized by citrate ions, making it effective in combating bacteria and other microorganisms. Due to its ability to inhibit the growth of bacteria and other microorganisms, silver citrate is frequently used in hygiene formulations, medical preparations, and specialty chemical applications.
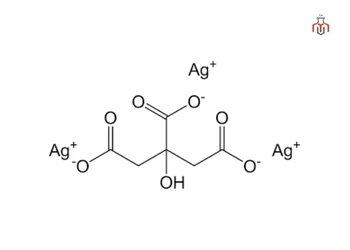
CAS No.: 126-45-4
Synonyms: Trisilver citrate, Citrate silver salt, Citric acid trisilver salt
| Physical Properties | |
| IUPAC Name | trisilver;2-hydroxypropane-1,2,3-tricarboxylate |
| Molecular Formula | C6H5Ag3O7 |
| Molecular Weight | 512.70 g/mol |
| Appearance (Form) | Solid Powder |
| Appearance (Color) | Off-White to Dark Grey |
| Solubility (In water) | 60.1 mg/mL |
| Pictograms : |  |
| Hazard Statements : | H315 Causes skin irritation |
| Precautionary statements: | P302+P352 IF ON SKIN: wash with plenty of water. P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present and easy to do – continue rinsing. P264 Do not breathe fume. P261 Avoid breathing dust |
Silver citrate is a silver salt of citric acid, stabilized by citrate ions, making it effective in combating bacteria and other microorganisms. Due to its ability to inhibit the growth of bacteria and other microorganisms, silver citrate is frequently used in hygiene formulations, medical preparations, and specialty chemical applications.

CAS No.: 126-45-4
Synonyms: Trisilver citrate, Citrate silver salt, Citric acid trisilver salt
| Physical Properties | |
| IUPAC Name | trisilver;2-hydroxypropane-1,2,3-tricarboxylate |
| Molecular Formula | C6H5Ag3O7 |
| Molecular Weight | 512.70 g/mol |
| Appearance (Form) | Solid Powder |
| Appearance (Color) | Off-White to Dark Grey |
| Solubility (In water) | 60.1 mg/mL |
| Pictograms : |  |
| Hazard Statements : | H315 Causes skin irritation |
| Precautionary statements: | P302+P352 IF ON SKIN: wash with plenty of water. P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present and easy to do – continue rinsing. P264 Do not breathe fume. P261 Avoid breathing dust |
Silver citrate is used in personal care products, medical devices, food processing, and cleaning products due to its antimicrobial and preservative properties.
Silver citrate is generally safe for skin when used in appropriate concentrations. It is commonly found in personal care products as a preservative and antimicrobial agent. However, individuals with sensitive skin should perform a patch test before use.
Silver citrate is antibacterial as it contains Ag⁺ ions in its composition, commonly used in the pharmaceutical, medical, oral care, and disinfectant industries as an antimicrobial agent.
Silver citrate has limited solubility in water. However, it can form functional antimicrobial systems in aqueous formulations when properly stabilized or combined with suitable formulation components.
Silver Citrate functions primarily as an antimicrobial agent due to the presence and controlled release of silver ions (Ag⁺). It works by releasing silver ions, which disrupt microbial DNA, proteins, and cell membranes, ultimately leading to the death of the microbes.