
Dimethylglyoxime, also known as DMG, is a chemical compound with the molecular formula C4H8N2O2. It is a diamide i.e., containing two amide functional groups in its structure. The neutral and anionic forms of this substance are denoted by the acronyms dmgH2 and dmgH−, respectively, where H represents hydrogen. It is a well known reagent in analytical chemistry.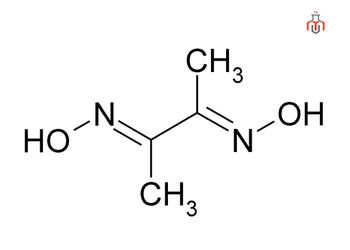
CAS No.: 95-45-4
Synonyms: Biacetyl dioxime; Diacetyldioxime; Chugaev’s reagent; 2,3-Diisonitrosobutane; Butane-2,3-dioxime
| Physical Properties | |
| Molecular formula | C4H8N2O2 |
| IUPAC Name | N,N′-Dihydroxy-2,3-butanediimine |
| Molecular weight | 116.12 g/mol |
| Chemical Properties | |
| Appearance (form) | Crystalline Solid Powder |
| Appearance (color) | White to Off-white |
| Solubility | Soluble 2% in Methanol |
| Density | 1.37 g/cm3 |
| Melting Point | 238-242°C (dec.) |
| Sulphated Ash | NMT 0.1% |
| Assay (Via Nickel complex) | NLT 98.0% |
Dimethylglyoxime has several uses in analytical chemistry, some of which are as follows-
| Pictograms : |   |
| Hazard Statements : | H228 – Flammable solid. |
| Precautionary statements : | P210 – Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking. P301+P310 – IF SWALLOWED: Immediately call a POISON CENTER or doctor. |
Dimethylglyoxime, also known as DMG, is a chemical compound with the molecular formula C4H8N2O2. It is a diamide i.e., containing two amide functional groups in its structure. The neutral and anionic forms of this substance are denoted by the acronyms dmgH2 and dmgH−, respectively, where H represents hydrogen. It is a well known reagent in analytical chemistry.
CAS No.: 95-45-4
Synonyms: Biacetyl dioxime; Diacetyldioxime; Chugaev’s reagent; 2,3-Diisonitrosobutane; Butane-2,3-dioxime
| Physical Properties | |
| Molecular formula | C4H8N2O2 |
| IUPAC Name | N,N′-Dihydroxy-2,3-butanediimine |
| Molecular weight | 116.12 g/mol |
| Chemical Properties | |
| Appearance (form) | Crystalline Solid Powder |
| Appearance (color) | White to Off-white |
| Solubility | Soluble 2% in Methanol |
| Density | 1.37 g/cm3 |
| Melting Point | 238-242°C (dec.) |
| Sulphated Ash | NMT 0.1% |
| Assay (Via Nickel complex) | NLT 98.0% |
Dimethylglyoxime has several uses in analytical chemistry, some of which are as follows-
| Pictograms : |   |
| Hazard Statements : | H228 – Flammable solid. |
| Precautionary statements : | P210 – Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking. P301+P310 – IF SWALLOWED: Immediately call a POISON CENTER or doctor. |
Dimethylglyoxime has many applications in analytical chemistry, which includes as a detecting reagent, a precipitating reagent, and a photometric reagent for various metal ions such as platinum, palladium and nickel.
Dimethylglyoxime may cause irritation upon inhalation, ingestion or upon coming in contact with eye/skin. It is also flammable under certain conditions and thus needs to be handled with care.
Dimethylglyoxime (DMG) is a weak acid.