
Methyl Green is a cationic (basic) triphenylmethane dye widely used in biological, diagnostic, and industrial applications. It is used in combination with Pyronin Y to achieve differential staining of biological specimens, distinguishing DNA in the nucleus from RNA in the cytoplasm. It is also used as a pH indicator, a counterstain in enzyme histochemistry, a tracking dye in gel electrophoresis, and a model cationic dye in wastewater treatment studies.
CAS No.: 7114-03-6
Synonyms: Basic Blue 20; Methyl green chloride
| Physical Properties | |
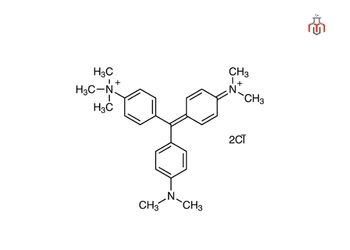
| Chemical formula | C26H33Cl2N3 |
| IUPAC Name | [4-[[4-(dimethylamino)phenyl]-(4-dimethylazaniumylidenecyclohexa -2,5-dien-1-ylidene)methyl]phenyl]-trimethylazanium dichloride |
| Molecular weight | 458.5 g/mol |
| Solubility | Soluble in Water and alcohol |
| Color | Dark green to black |
| Apperance | powder, or crystalline form |
| Pictograms : | |
| Hazard Statements : | H314 Causes severe skin burns and eye damage |
| Precautionary statements : | P273 Avoid release to the environment P302+P352 IF ON SKIN: Wash with plenty of water |
Methyl Green is a cationic (basic) triphenylmethane dye widely used in biological, diagnostic, and industrial applications. It is used in combination with Pyronin Y to achieve differential staining of biological specimens, distinguishing DNA in the nucleus from RNA in the cytoplasm. It is also used as a pH indicator, a counterstain in enzyme histochemistry, a tracking dye in gel electrophoresis, and a model cationic dye in wastewater treatment studies.

CAS No.: 7114-03-6
Synonyms: Basic Blue 20; Methyl green chloride
| Physical Properties | |
| Chemical formula | C26H33Cl2N3 |
| IUPAC Name | [4-[[4-(dimethylamino)phenyl]-(4-dimethylazaniumylidenecyclohexa -2,5-dien-1-ylidene)methyl]phenyl]-trimethylazanium dichloride |
| Molecular weight | 458.5 g/mol |
| Solubility | Soluble in Water and alcohol |
| Color | Dark green to black |
| Apperance | powder, or crystalline form |
| Pictograms : | |
| Hazard Statements : | H314 Causes severe skin burns and eye damage |
| Precautionary statements : | P273 Avoid release to the environment P302+P352 IF ON SKIN: Wash with plenty of water |
Methyl green is primarily used as a biological stain for nucleic acids, with a strong affinity for DNA. It is widely applied in histology and cytology to selectively stain cell nuclei, making it valuable for studying cell structure and tissue morphology.
Methyl green–pyronin Y stain is used to differentiate between DNA and RNA within cells. Methyl green stains DNA (typically green or blue-green), while pyronin Y stains RNA (red). This dual-staining technique is especially useful for identifying cellular activity, such as protein synthesis, and distinguishing different cell types based on nucleic acid content.
Methyl green is applied in microscopy and histological studies to visualize DNA distribution within cells. It plays a key role in analyzing nuclear structure, detecting abnormalities, and supporting research in cell biology, pathology, and genetics.
Methyl Green is a cationic dye used in laboratory staining and may cause serious eye damage, skin irritation, and respiratory irritation. It can be handled and used appropriately in controlled laboratory or industrial settings.