

Ethidium bromide (EtBr) is a phenanthridinium dye, widely used in molecular biology as a fluorescent tag (nucleic acid stain). It is particularly valuable for detecting DNA and RNA in techniques such as agarose gel electrophoresis and polyacrylamide gel electrophoresis (PAGE). Ethidium bromide binds to nucleic acids by intercalating between adjacent base pairs of DNA or RNA, and exhibits enhanced fluorescence, allowing nucleic acid bands to be visualized under ultraviolet (UV) light.
CAS No.: 1239-45-8
Synonyms: Homidium Bromide;Homidii bromidum; Bromuro de homidio; Bromure d’homidium; EtBr
| Physical Properties | |
| Chemical formula | C21H20BrN3 |
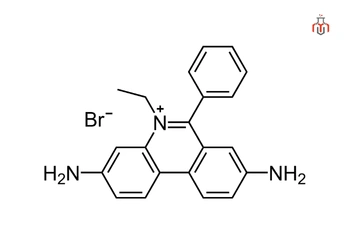
| IUPAC Name | 3,8-diamino-5-ethyl-6-phenylphenanthridin-5-ium bromide |
| Molecular weight | 394.3 g/mol |
| Solubility | Soluble in water, slightly soluble in ethanol, and partially soluble in chloroform |
| Flash Point | >100 °C |
| Density | 1.3739 |
| Chemical Properties | |
| Colour | Purple-red |
| State | Crystalline powder |
| Melting point | 260-262 °C (Decomposes) |
| Odor | Odorless |
| pH | 4.4 (20°C, 10g/L in H2O) |
| Pictograms : | |
| Hazard Statements : | H302 Harmful if swallowed |
| Precautionary statements : | P201 Obtain special instructions before use |
Ethidium bromide (EtBr) is a phenanthridinium dye, widely used in molecular biology as a fluorescent tag (nucleic acid stain). It is particularly valuable for detecting DNA and RNA in techniques such as agarose gel electrophoresis and polyacrylamide gel electrophoresis (PAGE). Ethidium bromide binds to nucleic acids by intercalating between adjacent base pairs of DNA or RNA, and exhibits enhanced fluorescence, allowing nucleic acid bands to be visualized under ultraviolet (UV) light.

CAS No.: 1239-45-8
Synonyms: Homidium Bromide;Homidii bromidum; Bromuro de homidio; Bromure d’homidium; EtBr
| Physical Properties | |
| Chemical formula | C21H20BrN3 |
| IUPAC Name | 3,8-diamino-5-ethyl-6-phenylphenanthridin-5-ium bromide |
| Molecular weight | 394.3 g/mol |
| Solubility | Soluble in water, slightly soluble in ethanol, and partially soluble in chloroform |
| Flash Point | >100 °C |
| Density | 1.3739 |
| Chemical Properties | |
| Colour | Purple-red |
| State | Crystalline powder |
| Melting point | 260-262 °C (Decomposes) |
| Odor | Odorless |
| pH | 4.4 (20°C, 10g/L in H2O) |
| Pictograms : | |
| Hazard Statements : | H302 Harmful if swallowed |
| Precautionary statements : | P201 Obtain special instructions before use |
Ethidium Bromide is used in DNA fragmentation by Agarose gel electrophoresis to make the DNA bands visible when the agarose gel is illuminated using UV light.
In agarose gel electrophoresis, Ethidium Bromide (EtBr) is typically used at a final concentration of about 0.2 to 0.5 microgram/ml in the gel.
Ethidium Bromide interacts with DNA by intercalating between adjacent base pairs in the DNA double helix. This interaction increases its fluorescence when exposed to UV light, which enables the detection of DNA fragments during gel electrophoresis.
Ethidium bromide is not radioactive. It is a fluorescent dye used for nucleic acid staining. However, it is advisable to check the safety information before handling it, as it is considered a mutagenic compound and should be handled with appropriate laboratory safety precautions.